Yamanashi Univ: Developing platinum-cobalt alloy / hydrogen electrode catalyst: drastically reducing fuel cell deterioration
January 14, 2020
NEDO / Yamanashi University / Tanaka Kikinzoku Kogyo
NEDO is jointly developing an innovative electrode catalyst / electrolyte membrane for FCV fuel cells.
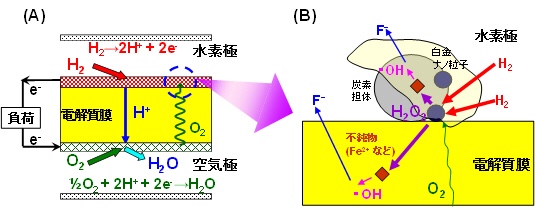
Conventionally, the generation of hydrogen peroxide (H2O2) has been the cause of hydrogen electrode / electrolyte membrane deterioration in polymer electrolyte fuel cells.
In recent years, we have succeeded in developing the world’s first platinum-cobalt alloy / hydrogen electrode catalyst that reduces the generation of hydrogen peroxide (H2O2) to less than half.
Platinum-cobalt alloy: hydrogen electrode catalyst
The newly developed platinum-cobalt alloy: hydrogen electrode catalyst has been incorporated into the fuel cell.
As a result, the durability of the electrolyte membrane is more than four times higher than that of conventional commercial / platinum hydrogen electrode catalysts.
It is expected that the durability of FCVs and stationary fuel cells will improve dramatically in the future.
Functional overview:
This platinum-cobalt alloy: hydrogen electrode catalyst greatly reduces the rate of hydrogen peroxide (H2O2: decomposition and degradation of the electrolyte membrane).
Due to the H2O2 generation rate / suppression effect of this catalyst, “PtCo / CHT catalyst mass synthesis * 4″ has become possible.
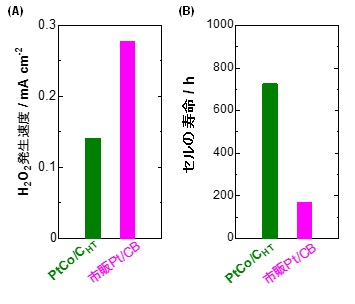
Confirmed that “H2O2 generation rate of PtCo / CHT catalyst is suppressed to less than half compared to commercial Pt / CB catalyst” (Fig. 1 (A))
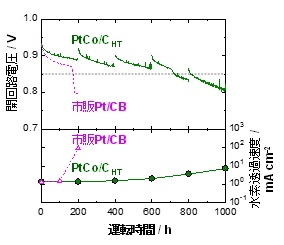
Conducted “accelerated deterioration test of single fuel cell * 5” using “electrolyte membrane using PtCo / CHT catalyst as hydrogen electrode”.
Confirmed that “durability is improved 4 times or more compared to commercial Pt / CB catalyst” (Fig. 1 (B))
The results of this research were published online on January 9, 2020, in the Royal Society of Chemistry (RSC) Journal of Materials Chemistry A.