COVID-19:Takara Bio PCR测试,在2小时内进行5000次:紧急申请美国FDA
COVID-19:
Takara Bio美国:
6月9日,美国一家子公司表示:“我们已经开发出一种方法,可以大大提高针对新冠状病毒感染的PCR检测效率。”
2小时内最多可以检查5,000个案件。
他正在向美国FDA申请紧急使用许可证,预计将在6月底获得批准。
新的PCR测试方法:
它是由Takara Bio的美国子公司(Takara Bio USA)和bioSyntagma通过结合现有设备和试剂共同开发的。
与美国目前的主流方法相比,它可以大大加快检查速度。
BioSyntagma拥有此方法的权利。
Takara Bio说:“我不打算在日本扩大它。”
京都新闻
https://www.kyoto-np.co.jp/articles/-/272636
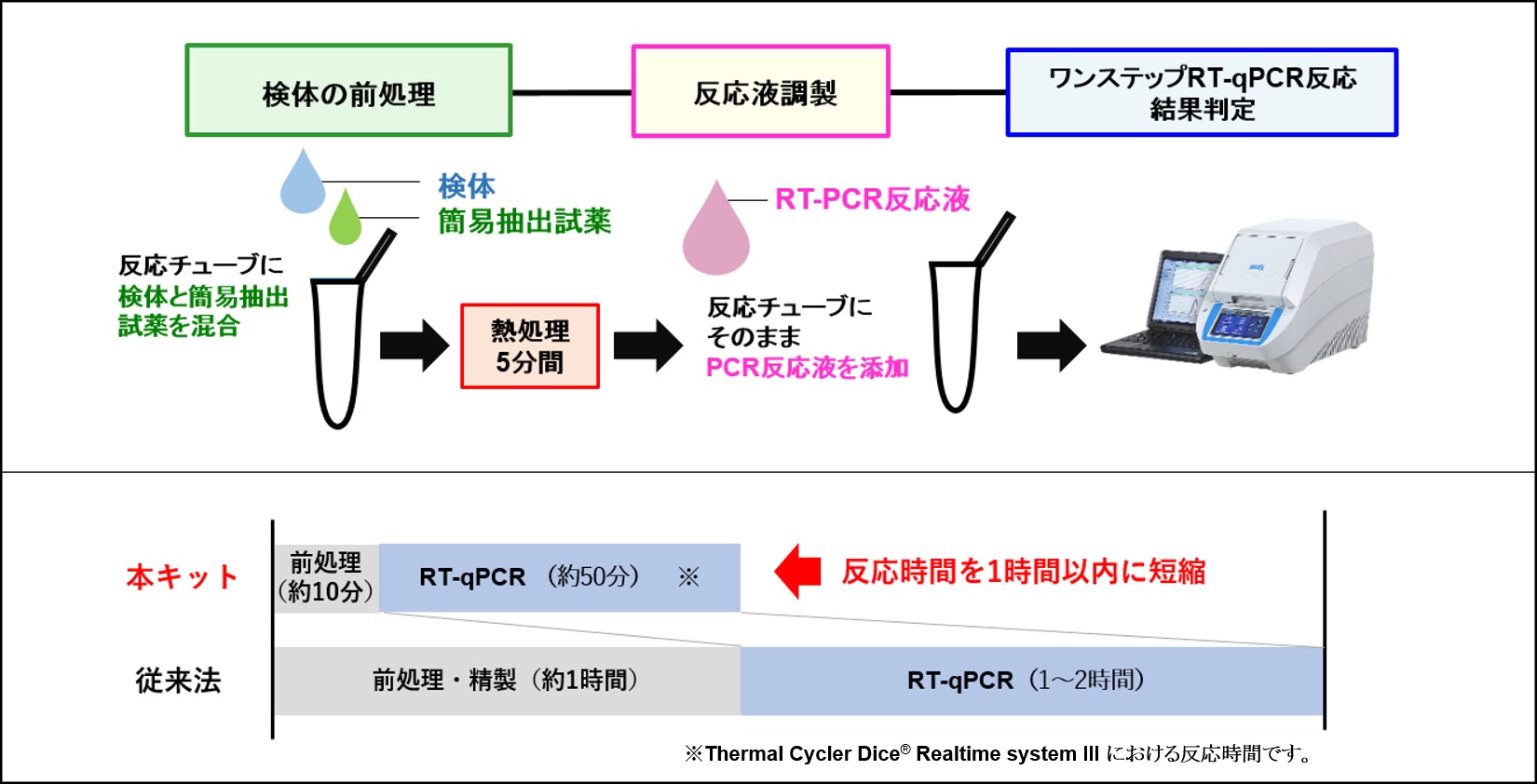
新型冠状病毒高通量PCR检测方法的开发
Takara Bio USA,Inc.,Takara Bio Inc.的美国子公司(美国加利福尼亚州TB USA)
2020年6月8日(当地时间),我们发布了新闻稿,介绍了针对新冠状病毒的高通量PCR检测方法的开发。
https://ir.takara-bio.co.jp/ja/news_all/news_IR/auto_20200610441125/pdfFile.pdf
Takara Bio USA, Inc. and bioSyntagma, Inc. develop method for large-scale automated COVID-19 testing
DATE: June 8, 2020
AUTHOR: Takara Bio USA, Inc.
CATEGORIES: Press release
Mountain View, CA—
June 8, 2020—
Takara Bio USA, Inc. (TBUSA), a pioneering life science instrument and reagent company and wholly owned subsidiary of Takara Bio Inc.,
collaborated with bioSyntagma, Inc. and their partners to develop and validate a new high-throughput method for detecting SARS-CoV-2.
The method
employs automation technology and reagents from TBUSA to detect viral RNA via real-time PCR and will enable rapid, large-scale testing of thousands of patient samples per day.
The method uses TBUSA’s SmartChip real-time PCR instrument, chips, and reagents to run 5,184 reactions per chip in less than 30 minutes of direct hands-on time.
Each reaction is at nanoliter scale, which reduces variability via elimination of the standard preamplification step and reduces costs via decreased reagent volume.
The trusted SmartChip Real-Time PCR System is already widely used for detection of antibiotic and antimicrobial resistance around the world, and is ideally suited to address the major need for rapid and accurate SARS-CoV-2 detection.
President of TBUSA Carol Lou states,
As shelter-in-place orders are lifted, controlling the COVID-19 pandemic will depend on our ability to detect SARS-CoV-2 from a large number of samples with precision, reproducibility, and speed.
To support this effort,
we optimized our existing chemistries and developed SmartChip protocols that maximize the number of samples processed while minimizing costs.
The work we have accomplished with bioSyntagma, plus their partners’ further development of diagnostic tests based on our work, will contribute to comprehensive and faster detection of COVID-19.
Scottsdale-based bioSyntagma
is a biotech spinoff of Arizona State University and serves as the development and validation partner of P2 Diagnostics, LLC.
The new SmartChip testing method
will be adopted by this and other molecular testing labs that are certified by CLIA (Clinical Laboratory Improvement Amendments) and therefore eligible to develop and perform COVID-19 diagnostic tests.
“The rapid development of this novel COVID-19 detection method
was made possible through a highly productive collaboration between bioSyntagma and TBUSA,
and the results of this effort will soon have an impact on the ability to detect SARS-CoV-2 in nasal and saliva samples from many patients,
said David Richardson, CEO of bioSyntagma.
bioSyntagma and partners
are seeking an Emergency Use Authorization (EUA) from the FDA for COVID-19 detection using the TBUSA SmartChip method.
Ipsum Diagnostics, LLC and Hackensack University Medical Center—two of TBUSA’s customers in the US—
have already obtained EUAs for their COVID-19 tests using Takara Bio’s one-step RT-PCR reagents.